
 Mendeleev vigorously opposed Meyer's method for representing periodicity with a graphic curve. He argued that graphs implied that there were no "breaks" between elements, or that there may be an indefinite number of chemical elements.1
Mendeleev vigorously opposed Meyer's method for representing periodicity with a graphic curve. He argued that graphs implied that there were no "breaks" between elements, or that there may be an indefinite number of chemical elements.1

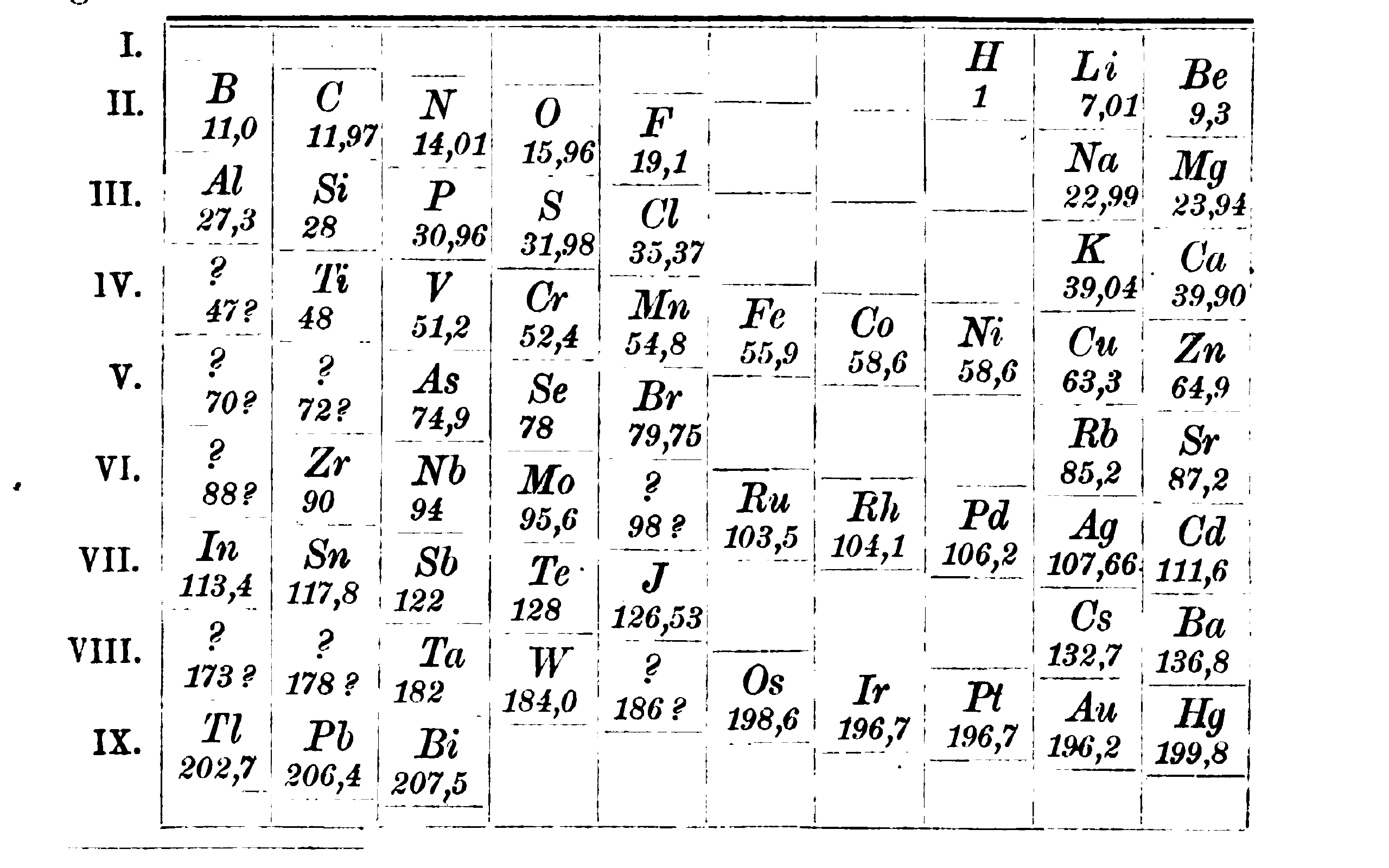
Meyer’s later investigations reflect his philosophical views about the relationship between theory and experiment. After the publication of the curve, Meyer re-determined the atomic weights of elements together with Karl Seubert. This work culminated in Die Atomgewichte Der Elemente Aus Den Originalzahlen Neu Berechnet (1883). Above, we can see how Meyer and Seubert included scandium (ekaboron).2
